How to Read a Peptide COA (Certificate of Analysis): A Simple Guide for Canadian Buyers
A Certificate of Analysis, usually called a COA, is one of the most important documents you can review before trusting any peptide product page. In regulated pharmaceutical manufacturing, a COA is part of the batch analysis record and summarizes what was tested, how it was tested, and how the batch compared with its specifications. In FDA guidance for active pharmaceutical ingredients, certificates of analysis are expected to include the product name, batch number, date of release, and information about the manufacturer, along with testing and reference details.
For Canadian buyers, the COA matters for another reason, too: not every product sold online is authorized for sale in Canada, and Health Canada states that only health products authorized for sale may be advertised in Canada. Health Canada also says that when a product is offered for sale in Canada to treat or prevent diseases or symptoms, it is regulated as a drug under the Food and Drugs Act. That makes the COA useful, but not sufficient on its own. A document can look professional and still not prove that a product is authorized or appropriate for the claims being made about it. google

What a COA is supposed to tell you
A real COA should help you answer five basic questions: what the material is, which batch it came from, who issued the document, what was tested, and whether the results matched the stated specifications. FDA guidance says authentic certificates of analysis should be issued for each batch of intermediate or API on request, and that the certificate should show the material name, batch number, and date of release. It should also show the name and address of the original manufacturer, or the repacker/reprocessor if that is who performed the analysis.
That is the first thing to understand about reading a peptide COA. The document is not mainly about marketing. It is about traceability and testing. If a supplier is serious about quality, the COA should let you connect the paper to a specific batch, a specific test set, and a specific source. If it does not do that, the document is much less useful.

Start with the product name and batch number
The first line to check is the product name. It should match the product you are looking at on the website exactly or very closely. Then check the batch number, lot number, or control number. FDA guidance says the batch identity should be included in batch analysis reports, and the COA should show the batch number and date of release. Without that connection, a COA may be generic rather than product-specific.
A batch number matters because it lets you know whether the document belongs to the exact material you are reviewing. If the COA does not match the product name, batch number, or packaging label, you should treat that as a warning sign. A trustworthy supplier should make it easy to cross-check the website, the label, and the certificate.
Check who issued the COA
The next item to look for is the issuer. FDA guidance says the COA should show the name, address, and telephone number of the original manufacturer, and if the analysis was done by a repacker or reprocessor, that should also be clearly stated along with a reference to the original manufacturer. That detail is useful because it tells you who is responsible for the analysis.
If the document has no issuer name, no contact information, or no link to the manufacturer, it is harder to judge reliability. A proper COA should not feel anonymous. It should tell you who performed or issued the analysis and make that source traceable.
Look for the date of release, and sometimes expiry or retest dates
A good COA should show the date the batch was released. For some materials, especially APIs, FDA guidance also says the label and COA should show an expiry date or retest date where applicable. That matters because a test result from years ago may not tell you much about the current condition of the material.
If you see a COA with no date, or one where the date is far removed from the batch being sold, be careful. For a quality-focused listing, the date should help you understand how recent the analysis is and whether the document still makes sense for the product being offered.
Read the test results, not just the headline claims
This is where many people stop too early. Some websites highlight one big number, often purity, while hiding the rest of the data. FDA guidance says batch analysis reports should include results from all tests performed on the batch, including tests not part of the proposed specification, and results should be expressed numerically or qualitatively as appropriate. The guidance also discourages vague terms such as “conforms” or “meets specification.”
That means a strong COA should show more than a single polished headline. You should look for the actual test list, the method references, and the numerical values or clear qualitative results. If the document only says “passes” or “meets spec” without showing the real data, it gives you less to evaluate.
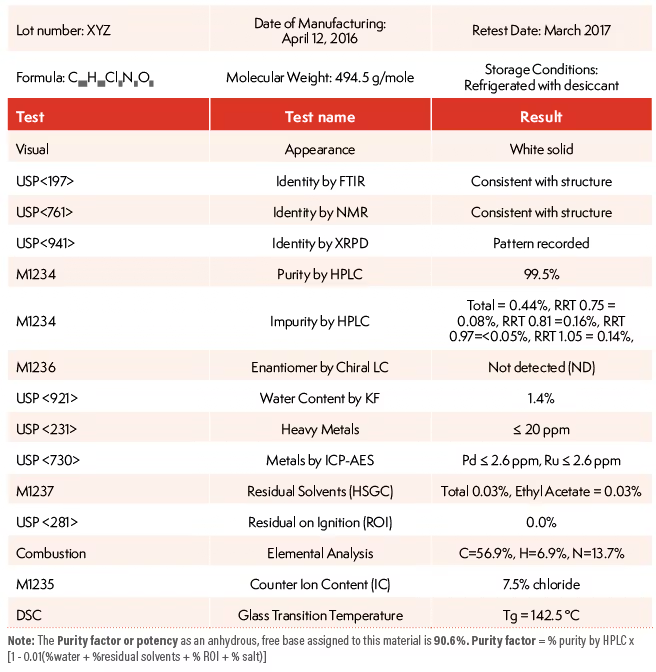
What tests commonly appear on a peptide COA
Depending on the product, a peptide COA may include identity testing, assay or purity testing, impurity analysis, and sometimes other quality checks. FDA guidance for drug substances emphasizes that batch reports should include tests performed on the batch and references to analytical procedures. It also notes that specifications and batch analyses are used to evaluate manufacturing consistency.
For peptides specifically, the most relevant items are usually the ones that show whether the material matches its stated identity and whether any impurities are present. In practice, that is why buyers often focus on purity and identity testing. A COA that leaves out method references, omits batch-specific testing, or hides the impurity information is less informative than one that gives the full picture.
Pay attention to the impurity section
Impurity data is one of the most important parts of the document. FDA guidance says specifications for intermediates should usually include testing for assay and impurities, and batch analysis reports should include all tests performed on the batch. The point is not just to say that a material was made, but to show what else may be present in it.
A strong COA should therefore make impurity information easy to find and easy to understand. If a document glosses over impurities or buries them in vague language, that is a reason to slow down and inspect the rest of the listing more carefully. A supplier that is comfortable with quality usually has no problem showing this section clearly.
Check whether the analytical method is listed
A COA is only as meaningful as the method behind it. FDA guidance says the batch analysis report should include references to analytical procedures, and in the FDA’s guidance on drug substances, the specification sheet should identify the tests and the procedures used. When multiple analytical procedures are possible, the document should explain which methods were used and what acceptance criteria applied.
For a buyer, this means the method section helps you understand how the result was generated. A real COA is not just a photo of a result sheet. It should also tell you which test method produced that result, because the method is part of the evidence.
Do not confuse a COA with authorization
This is one of the most important distinctions for a Canadian website. A COA may show that a batch was tested, but it does not prove the product is authorized for sale in Canada. Health Canada says only health products authorized for sale in Canada may be advertised, and it also says that products offered for sale in Canada to treat or prevent diseases or symptoms are regulated as drugs.
Health Canada has also issued warnings about unauthorized injectable peptide drugs sold online, noting that these products may be misrepresented and have not been assessed for safety, efficacy, and quality. That is why a buyer should treat the COA as one piece of evidence, not the final answer. The COA can support a quality discussion, but it cannot by itself convert an unauthorized product into an authorized one.
Signs that a peptide COA may be weak or unreliable
A weak COA often has the same patterns: it is generic, it lacks batch numbers, it omits the issuer, it shows no dates, or it uses vague wording instead of actual results. FDA guidance specifically discourages “conforms” and “meets specification” as the main way to report results, because those phrases do not show the underlying data.
Another red flag is a certificate that looks polished but cannot be tied back to the product you are viewing. If the website, the label, and the COA do not line up, the document is not doing its job. A serious supplier should make the chain of information easy to verify.

What “good” looks like on a COA
A strong COA is usually simple, complete, and consistent. It identifies the material clearly, shows the batch number, includes the release date, names the manufacturer or issuer, lists the tests performed, references the analytical method, and gives clear results. FDA guidance also notes that batch analysis reports should include a description of the batch and the tests performed, with results presented numerically or qualitatively as appropriate.
That is the standard to aim for when you review a peptide listing. The best documents are not flashy. They are organized, traceable, and easy to match to the exact item you are considering. That is what gives the COA real value.
Why this matters for Canadian buyers
For a Canadian audience, the stakes are higher because product advertising and product status both matter. Health Canada is explicit that advertising must be compliant, and only authorized products may be advertised in Canada. If a peptide page is built around claims, it can create problems even before you get to the document.
That is why the smartest way to use a COA is as part of a broader review. Read the website description, inspect the batch data, compare the product page with the certificate, and make sure nothing conflicts. A COA should support transparency, not replace it.
A simple way to review a COA step by step
Start with the top of the document and check the product name. Then confirm the batch or lot number. After that, identify the manufacturer or issuer and check the date. Once those basics line up, move to the test list and the results. Finally, review the impurity information, the method references, and any expiry or retest details that apply. That sequence follows the way FDA guidance structures batch analysis reports and certificates of analysis.
If all of those items make sense and match the product page, you have a much stronger basis for trust. If any of them fail, do not assume the rest of the document is reliable just because it looks professional. In quality review, consistency is everything.

Final thoughts
Reading a peptide COA is really about learning how to separate appearance from evidence. A certificate should connect a product to a batch, a manufacturer, a test method, and a clear result set. In FDA guidance, that is exactly what batch analysis reports and certificates of analysis are meant to do. In Canada, that kind of transparency is even more important because Health Canada regulates health product advertising and unauthorized products can still circulate online.
If you remember only one thing, make it this: a COA should help you verify the specific batch in front of you. It should not be generic, vague, or disconnected from the product page. When the document is clear, the product presentation is clearer too.
⸻
FAQ
What does COA mean on a peptide product page?
COA means Certificate of Analysis. In industry guidance, it is a batch analysis document that shows what was tested, how it was tested, and how the batch compared with its specifications.
Should a peptide COA include the batch number?
Yes. FDA guidance says the batch identity, including the batch number, should be included on the certificate of analysis and in batch analysis records.
Is a COA the same as legal authorization in Canada?
No. Health Canada says only health products authorized for sale in Canada may be advertised, and a COA alone does not prove authorization.
What is the biggest red flag on a COA?
A common red flag is vague wording without real test data, especially if the document only says “conforms” or “meets specification” instead of showing numeric or clear qualitative results.
Why is the impurity section important?
Because it helps show what else may be present in the batch. FDA guidance says specifications and batch analysis reports should address assay and impurities, and the report should include results for the tests performed.
Should a COA show who performed the analysis?
Yes. FDA guidance says the certificate should identify the manufacturer or, if applicable, the repacker or reprocessor, along with contact details and a reference to the original manufacturer.
