Peptide Storage Guide: Temperature, Light, and Handling Basics
Peptides are sensitive materials, and how they are stored can affect whether they remain usable, stable, and consistent with the product information provided by the supplier. In the scientific literature, peptide stability is influenced by both intrinsic and external factors, and reviews of therapeutic peptides repeatedly point to temperature, light, humidity, oxidation, and handling as important stressors.
For Canadian buyers, the most important rule is simple: follow the storage conditions on the label or product page, because Health Canada says drugs should be stored according to the conditions described on the label and that any controls for temperature, humidity, and light need to be in place. That same guidance also says storage areas should be clean, dry, well ventilated, and monitored so the required conditions are actually maintained.
This guide explains peptide storage in plain language. It focuses on the basics that matter most: temperature, light, humidity, handling, packaging, transport, and the mistakes that can quietly ruin a product before it is ever used. It is educational only and should not replace the instructions on the label or advice from a qualified professional. google

Why peptide storage matters
A peptide is not just a name on a label. It is a chemical structure that can change when exposed to the wrong environment. Scientific reviews describe peptides as sensitive to external stressors such as heat, light, pH, oxidation, and humidity, all of which can influence stability and performance.
That is why storage is not a small detail. If a product is kept under conditions that do not match the label, the issue is not only convenience; it can become a quality problem. FDA stability guidance explains that drug products may need special labeling restrictions when they are expected to encounter extremes of temperature, humidity, air, or light, and those restrictions should be supported by data.
For a website selling peptide-related products, this also matters for trust. Clear storage information is one of the easiest ways to show that a supplier understands product care and is not treating all peptides as interchangeable. Stability is product-specific, not one-size-fits-all.
Temperature: the first thing to get right
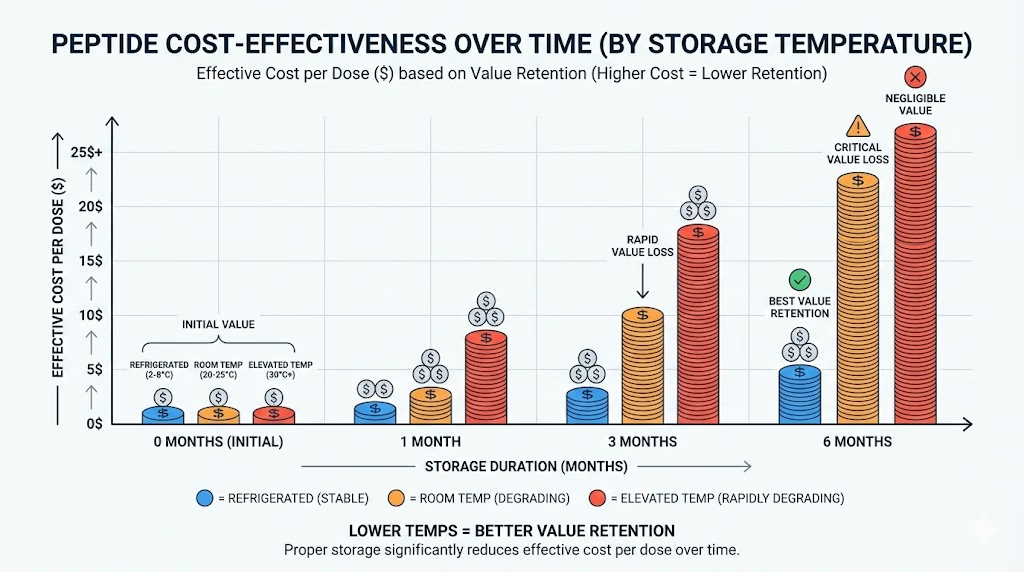
Temperature is usually the first storage condition people think about, and for good reason. Stability guidance from FDA and Health Canada both treat temperature as a major factor in storage and transport, and FDA guidance specifically discusses how drug substances and drug products may need different long-term, refrigerated, or frozen storage conditions depending on the product.
The practical takeaway is that you should never assume every peptide belongs in the same place. Some products are meant for refrigerated storage, others for room temperature, and others for freezing or even below freezing, depending on how they were designed and labeled. Stability guidance says the correct condition depends on the product and the available stability data, not on a generic rule that applies to every item.
Short excursions can also matter. FDA guidance notes that stability assessments should consider short-term excursions outside the label storage conditions, including during shipping or handling. In other words, a brief temperature change may or may not be acceptable, but it is something that should be evaluated rather than ignored.
For buyers, the best habit is to check three places: the product label, the product page, and any documentation supplied by the seller. If those sources disagree on temperature, that is a warning sign. If they match, storage becomes much easier to manage. Health Canada explicitly says products should be stored according to the label and that temperature controls should be in place wherever the product is stored.
Light: the hidden source of damage
Light is easy to overlook because it does not feel as dramatic as heat or freezing, but stability guidance treats light as a real stressor. FDA’s drug stability guidance specifically lists sunlight, ultraviolet light, and other artificial light among the conditions that can affect drug products, and says special label restrictions may be needed when exposure to those factors is expected.
Health Canada’s storage guidance also names light alongside temperature and humidity as a condition that may need to be controlled. That means the question is not only “Is the product cold enough?” but also “Is it protected from exposure?”
In practical terms, light protection means avoiding unnecessary exposure and following the instructions on the label. If a product says it should be protected from light, that instruction should be treated as part of the storage requirement, not as optional advice. Stability guidance says recommended restrictions should be practical and supported by appropriate data, which is another reminder that light protection is a real product issue, not just a packaging preference.
For a site owner, it is smart to present light guidance clearly on the product page. A buyer should not have to dig through a FAQ or support email to find out whether a product needs protection from light. If the label requires it, the listing should say so plainly.

Humidity and moisture: the condition people forget
Humidity is another factor that can affect storage even when temperature looks fine. FDA stability guidance lists humidity with temperature, air, and light as a factor that may require special restrictions, and Health Canada says controls for conditions such as humidity should be in place when they are specified on the label.
This matters because a product can be exposed to moisture during storage, transport, or repeated opening and closing of the container. The point is not that every peptide is equally sensitive in the same way, but that moisture control is part of the stability picture and should never be ignored simply because the product is stored indoors.
On a product page, this can be handled simply and safely. You do not need dramatic wording. A straightforward line such as “Store according to the label. Protect from conditions that may affect stability, including heat, moisture, and light where applicable” is clearer than vague marketing language. That approach matches the regulatory emphasis on label-based storage conditions.
Handling basics: every opening matters
Storage is not only about where a product sits. It is also about how it is handled. Every time a container is opened, moved, or exposed to the wrong environment, the risk of contamination or stress increases. Stability guidance repeatedly treats storage and handling together, which is why short excursions and label-based restrictions are discussed in the same documents.
The safest handling approach is the simplest one: open the container only when needed, avoid unnecessary exposure, and return it to the proper storage condition immediately. Health Canada’s guidance on warehousing and storage also stresses monitoring and good storage-area conditions, which reflects the same principle on the supply side.
Another useful habit is to keep the container and its documentation together. If a product has a lot number, batch number, or storage note, that information should remain easy to find. A good storage routine protects not just the material but the traceability of the material.
Shipping and short-term excursions
Shipping is one of the most common moments when products leave ideal conditions. FDA guidance specifically notes that stability studies should address the effect of short-term excursions outside the proposed label storage condition, including during shipping and handling. It also says alternative storage conditions can be used if justified by stability data.
That means shipping is not a minor detail to ignore. Even if a product is ultimately stored correctly after arrival, the transport phase still matters. For buyers, that is a strong reason to choose suppliers that explain how the product is packed, shipped, and protected during transit.
For site owners, this is one of the best trust signals you can publish. A product page or shipping page that tells buyers what to expect during transit, how the product is protected, and what storage condition should begin immediately after delivery is much more useful than a generic “shipped safely” claim. FDA stability guidance supports this kind of practical risk awareness.

Why packaging matters
Packaging is part of storage because the container helps determine what the product is exposed to. FDA guidance on stability notes that the effect of temperature, humidity, air, and light depends on how the product is packaged, and Health Canada guidance similarly expects storage conditions to be controlled in practice, not just written down.
For a peptide product, that means the seller should not treat the container as an afterthought. The packaging should support the stated storage condition, and the storage instructions should be compatible with how the product is shipped and handled. If a product requires special protection, the packaging and label should make that obvious.
Consumers do not need to become packaging experts, but they should notice when packaging and storage instructions seem inconsistent. A product advertised with careful temperature control but shipped in a way that does not match that claim deserves a closer look.
How to read storage instructions on a label
Storage instructions are part of the product information, not just legal fine print. Health Canada says products should be stored according to the conditions described on the label, and any required controls for temperature, humidity, or light should be in place.
When you read a label or product page, look for a few things: whether the storage condition is specific, whether it mentions light or moisture protection, whether it differs from room temperature, and whether the instruction is consistent across the product page and the packaging. If the label says one thing and the website says another, trust the more precise source and ask for clarification before acting.
It also helps to remember that “special restrictions” are normal in product science. FDA stability guidance says products exposed to extremes may require special labeling restrictions, and those restrictions should be practical and backed by data. So a product that has specific instructions is not automatically unusual; it may simply be showing that it was formulated with a defined stability profile.
The role of monitoring and consistency
Good storage is not only about one correct decision at purchase time. It is about consistency over time. Health Canada’s warehousing guidance says storage areas should be monitored, temperature should be controlled with calibrated devices, and records should be kept to show the conditions are being met.
That principle matters even for small-scale sellers and buyers because a product can be damaged by repeated exposure, not just one dramatic mistake. A stable-looking package can still be compromised if it sits in the wrong environment for too long or is repeatedly handled without care. Stability testing frameworks exist precisely because products can degrade over time and under stress.
For buyers, this means that a trustworthy seller should sound organized, not improvised. Clear product pages, readable storage notes, and consistent documentation are all signs that the seller understands how storage affects quality.
Common storage mistakes to avoid
One common mistake is assuming that every peptide should be stored in the same way. Stability guidance makes clear that conditions vary by product and by available data, so a one-size-fits-all approach is unsafe.
A second mistake is ignoring light exposure. Because FDA and Health Canada both treat light as a relevant condition, any instruction about protection from light should be taken seriously.
A third mistake is overlooking transport. The product may be stored correctly after delivery, but short-term excursions during shipping can still matter, and FDA guidance specifically calls out those excursions.
A fourth mistake is relying on vague seller language instead of label-based instructions. Health Canada’s guidance is explicit: store according to the label and ensure the stated conditions are actually in place. That is the standard to use.
What a good peptide storage page should say
A good product or educational page should be simple and useful. It should say that storage depends on the product label, that temperature, light, and humidity may matter, and that handling should minimize unnecessary exposure. It should not pretend that every product has the same requirements. That is consistent with the way stability guidance is written.
It is also helpful to include shipping and documentation notes on the same page or in closely linked pages. Buyers benefit when the site explains how the product is protected during transit and where to find the relevant label or batch information. That kind of structure follows the same traceability and monitoring mindset used in drug storage guidance.
Final thoughts
Peptide storage is really about respecting the product’s stability profile. Temperature matters. Light matters. Humidity matters. Handling matters. And the label matters most of all. Health Canada says drugs should be stored according to label conditions, while FDA stability guidance explains why products may need special restrictions and why short-term shipping excursions and environmental stressors have to be considered.
For Canadian buyers and site owners, the simplest rule is the best one: follow the product instructions, keep the storage environment consistent, and do not assume all peptides behave the same way. The more specific the storage information, the easier it is to protect product quality and build trust.


⸻
FAQ
Do all peptides need the same storage conditions?
No. Stability guidance makes clear that storage conditions depend on the product and the supporting stability data, so one product may require different conditions from another.
Is light really important for peptide storage?
Yes. FDA guidance specifically identifies sunlight, ultraviolet light, and other artificial light as stability factors, and Health Canada also lists light as a condition that may need control.
What should I trust first: the website or the label?
Trust the label first when there is any conflict. Health Canada says products should be stored according to the conditions described on the label.
Can shipping affect peptide quality?
Yes. FDA guidance explicitly discusses short-term excursions outside label storage conditions during shipping and handling.
Why do some peptide products have special storage restrictions?
Because exposure to temperature, humidity, air, and light can affect stability, and FDA guidance says such products may need practical, data-supported label restrictions.
What is the biggest storage mistake buyers make?
Assuming all peptides are stored the same way. Stability is product-specific, so buyers should always follow the exact instructions for the item they have.
